 
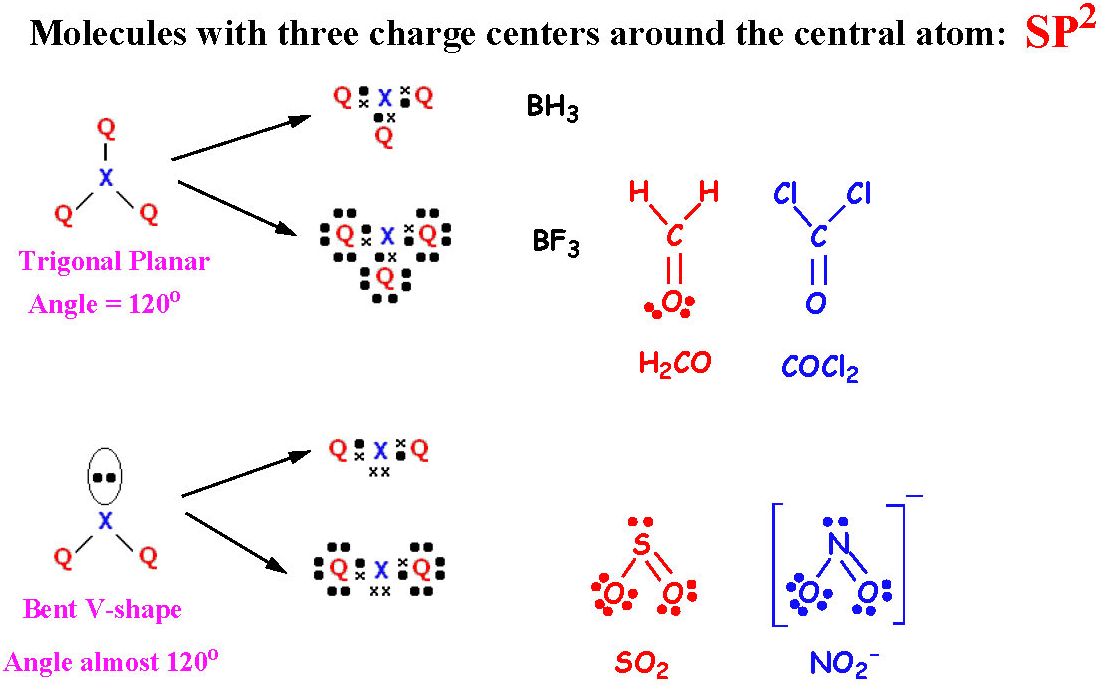
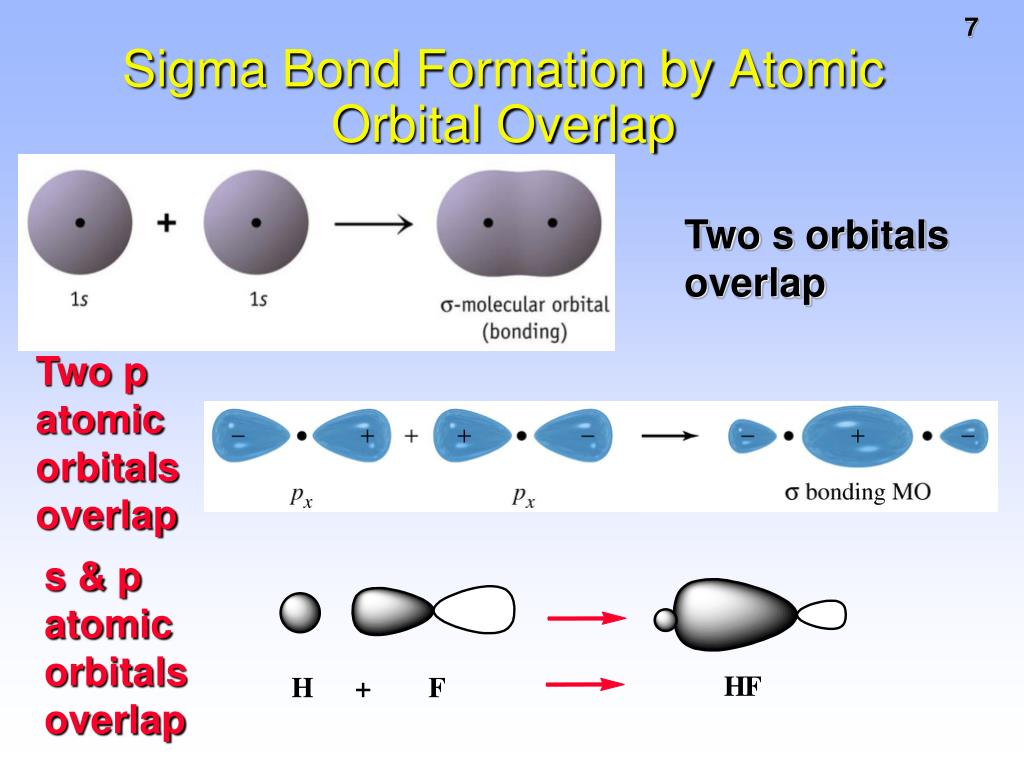
Valence electron pair repulsion (VSEPR) theory states that the geometry of the molecule depends on the repulsion of electron pairs in the valence shells.Įlectrons always tend to align themselves at the maximum distance from each other to minimize the repulsions as well as to make the arrangement stable. VSEPR theory helps in predicting the geometry and shape of covalently bonded molecules like BeI2. Hence, this is the most stable Lewis structure for BeI2. So, all the atoms in the BeI2 Lewis structure have zero formal charges.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
